Lead Halide Perovskites: Rainbow in a Bottle
Fields covered: Material Science, Nanomaterials, Chemistry
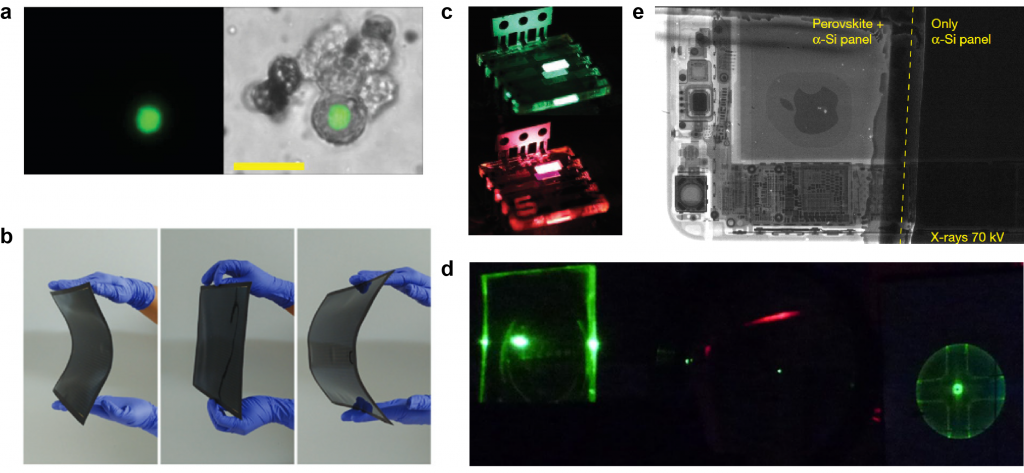
“Lead halide perovskites” has been keeping scientists on their toes for the past five years: not only can they be synthesized in large scales, at low costs and at room temperatures,6 they boast excellent optical properties such as high brightness and color purity.7 Lead halide perovskites has thus been shown in a wide variety of applications ranging bio-medical imaging,1 solar cells,2 LEDs,3 lasers4 and x-ray film,5 just to name a few (Figure 1).
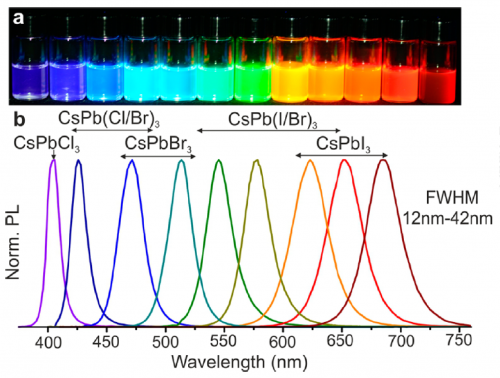
As its name suggests, lead halide perovskites are defined by their perovskite chemical formula APbX3, where A can be Cesium, Methylammonium or Formadinium Ammonium, Pb is lead, and X are the halides (e.g. chloride, bromide, and iodide). By controlling the halide composition, lead halide perovskites can be made to emit at any color throughout the visible range: perovskites rich in chloride, bromide and iodide each emits blue green and red respectively (Figure 2).7 Perhaps the most interesting aspect of halide perovskites is the ability to tune their emissions through halide exchange even after synthesis is complete; as an example, green emitting APbBr3 nanoparticles can be made to emit orange by adding an iodide source (e.g. iodotrimethylsilane).8 The ability to fine-tune the perovskites through such simple methods can enable engineers to make TVs with richer colors, or give bio-medical scientists a larger color palette for more precise bio-marking.
With the superb properties of lead halide perovskites, you might be wondering: why can’t I buy it in stores now? For one, lead halide perovskites are notoriously unstable to water and degrade in humid environments. This limits perovskites from reaching commercial benchmarks, such as deployment in solar cells for 15 years without voltage drop.9 Nevertheless, teams all across the world are applying a combination of strategies to inhibit contact with moisture, as well as improving the resistance of the crystal structure towards water damage. Further, to address the elephant in the room, lead halide perovskites contain lead, which can lead to brain damage when ingested. To address this, work is being done to substitute lead with cations such as manganese, bismuth and silver,6 as well as to introduce phosphates10 to entrap lead if leakage occurs.
Will the future of lead halide perovskites be as bright as their emissions, or will they be another over-promise the likes of graphene? If commercial startups at home and abroad are indicators to a field’s success, lead halide perovskites might see the light of day soon.
References:
Tan, M. J. H.; Ravichandran, D.; Ang, H. L.; Ong, E. W. Y.; Lim, C. Q. X.; Kam, G. M. Q.; Kumar, A. P.; Tan, Z.-K. Magneto-Fluorescent Perovskite Nanocomposites for Directed Cell Motion and Imaging. Adv. Healthc. Mater. 2019, 8 (23), 1900859. https://doi.org/https://doi.org/10.1002/adhm.201900859.
Chung, J.; Shin, S. S.; Hwang, K.; Kim, G.; Kim, K. W.; Lee, D. S.; Kim, W.; Ma, B. S.; Kim, Y. K.; Kim, T. S.; et al. Record-Efficiency Flexible Perovskite Solar Cell and Module Enabled by a Porous-Planar Structure as an Electron Transport Layer. Energy Environ. Sci. 2020, 13 (12), 4854–4861. https://doi.org/10.1039/d0ee02164d.
Tan, Z. K.; Moghaddam, R. S.; Lai, M. L.; Docampo, P.; Higler, R.; Deschler, F.; Price, M.; Sadhanala, A.; Pazos, L. M.; Credgington, D.; et al. Bright Light-Emitting Diodes Based on Organometal Halide Perovskite. Nat. Nanotechnol. 2014, 9 (9), 687–692. https://doi.org/10.1038/nnano.2014.149.
Harwell, J. R.; Whitworth, G. L.; Turnbull, G. A.; Samuel, I. D. W. Green Perovskite Distributed Feedback Lasers. Sci. Rep. 2017, 7 (1), 1–8. https://doi.org/10.1038/s41598-017-11569-3.
Chen, Q.; Wu, J.; Ou, X.; Huang, B.; Almutlaq, J.; Zhumekenov, A. A.; Guan, X.; Han, S.; Liang, L.; Yi, Z.; et al. All-Inorganic Perovskite Nanocrystal Scintillators. Nature 2018, 561 (7721), 88–93. https://doi.org/10.1038/s41586-018-0451-1.
Dey, A.; Ye, J.; De, A.; Debroye, E.; Ha, S. K.; Bladt, E.; Kshirsagar, A. S.; Wang, Z.; Yin, J.; Wang, Y.; et al. State of the Art and Prospects for Halide Perovskite Nanocrystals. ACS Nano 2021. https://doi.org/10.1021/acsnano.0c08903.
Protesescu, L.; Yakunin, S.; Bodnarchuk, M. I.; Krieg, F.; Caputo, R.; Hendon, C. H.; Yang, R. X.; Walsh, A.; Kovalenko, M. V. Nanocrystals of Cesium Lead Halide Perovskites (CsPbX3, X = Cl, Br, and I): Novel Optoelectronic Materials Showing Bright Emission with Wide Color Gamut. Nano Lett. 2015, 15 (6), 3692–3696. https://doi.org/10.1021/nl5048779.
Creutz, S. E.; Crites, E. N.; De Siena, M. C.; Gamelin, D. R. Anion Exchange in Cesium Lead Halide Perovskite Nanocrystals and Thin Films Using Trimethylsilyl Halide Reagents. Chem. Mater. 2018, 30 (15), 4887–4891. https://doi.org/10.1021/acs.chemmater.8b02100.
Meng, L.; You, J.; Yang, Y. Addressing the Stability Issue of Perovskite Solar Cells for Commercial Applications. Nat. Commun. 2018, 9 (1), 1–4. https://doi.org/10.1038/s41467-018-07255-1.
Rossi, L.; Mettan, X. Fighting Health Hazards in Lead Halide Perovskite Optoelectronic Devices with Transparent Phosphate Salts ̌. ACS Appl. Mater. Interfaces 2021, ASAP. https://doi.org/10.1021/acsami.0c21137.